Because the sun isn't always shining and the wind isn't always blowing, solar and wind power both need a way to store the renewable energy they produce. The obvious solution is batteries, but the lithium-ion (Li-ion) variety so essential to our phones and other portable devices are too expensive for the large scale required and are susceptible to combustion. Now, researchers have come up with a far cheaper and safer alternative with a creative approach to battery chemistries. Their work, which utilized high-brightness x-rays from the U.S. Department of Energy’s Advanced Photon Source (APS), was published in Nature.
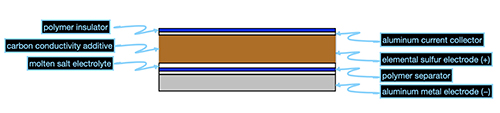
Attempts have been made to devise batteries with metal electrodes and without lithium, an element hampered by increasing shortages and growing ethical concerns surrounding its mining. But metal negative electrodes display a disquieting tendency over repeated cycling to form dendrites, thin needle-like filaments of metal that eventually grow to contact the other electrode and short out the battery. The new battery formulation conceived by these researchers employs an aluminum-chalcogen electrode chemistry combined with a molten salt electrolyte that not only avoids dendritic shorting but is based completely on elements that are among the most common on Earth and thus the cheapest: aluminum (Al), sulfur (S), and aluminum chloride-sodium chloride.
The researchers constructed and tested aluminum-chalcogen-based batteries in two different systems with aluminum anodes, one with a selenium (Se) cathode with an ionic liquid electrolyte and the other a sulfur cathode using a molten salt electrolyte and compared charge-transfer kinetics between the ionic liquid and molten salt electrolytes. The operando selenium reaction mechanism was investigated using x-ray absorption fine-structure spectroscopy at the APS X-ray Science Division Spectroscopy Group’s 20-BM-B beamline at the APS, a Department of Energy Office of Science user facility at Argonne National Laboratory.
Despite the usual preference for room-temperature ionic liquid electrolytes in rechargeable batteries, the team found that the aluminum-sulfur (Fig. 1) system with a molten chloroaluminate electrolyte displays a markedly better performance than the Al-Se battery. This includes not only faster charging but also extremely high cycling rates, with a capacity of 520 mAh g-1 over 100 cycles. The moderately elevated operating temperature of this Al|NaCl-KCl-AlCl3S battery is self-maintaining by the charge-discharge cycle, requiring no external temperature control; yet unlike Li-ion batteries, this battery system is immune to dangerous overheating and fire.
A rather serendipitous finding is that even at very high charging rates, the chloroaluminate salt electrolyte used in the aluminum-sulfur cell also proves to be the key in preventing dendritic shorting, apparently because its particular chemistry imposes a higher than usual threshold current for dendrite formation. This overcomes one of the former major obstacles to the use of metal negative electrodes.
The Al-S system demonstrated by the experimental team in this work proves to be more than simply an example of an intriguing battery design and chemistry. Because all of its components are readily available and inexpensive, it boasts an estimated cell-level cost of approximately $8.99 per kWh, a significant 12-16% reduction over contemporary Li-ion batteries. The battery can use a low-grade aluminum anode with little or no degradation in performance, and at larger scale does not require the active cooling essential for large Li-ion batteries. All of these characteristics, combined with the battery's resistance to fire, make it an excellent candidate for energy storage needs of some 10s of kilowatt hours, as in single homes or small businesses, and for electric vehicles, both onboard and in charging stations.
There's no doubt that Li-ion batteries are a well-established and successful technology, but they're not the only game in town. As the present work demonstrates, other innovative solutions to energy storage are not only possible but extremely economical and safe. These experiments may also point the way to further creative battery possibilities using other metals that may offer similar advantages for practical applications. ― Mark Wolverton
See: Quanquan Pang1*, Jiashen Meng1,2,3, Saransh Gupta4, Xufeng Hong1, Chun Yuen Kwok5, Ji Zhao3, Yingxia Jin3,6, Like Xu3, Ozlem Karahan3, Ziqi Wang3, Spencer Toll3, Liqiang Mai2, Linda F. Nazar5, Mahalingam Balasubramanian7, Badri Narayanan4, and Donald R. Sadoway3**, “Fast-charging aluminium–chalcogen batteries resistant to dendritic shorting,” Nature 608, 704 (25 August 2022). DOI: 10.1038/s41586-022-04983-9
Author affiliations: 1Peking University, 2Wuhan University of Technology, 3Massachusetts Institute of Technology, 4University of Louisville, 5University of Waterloo, 6Yunnan University, 7Argonne National Laboratory
Correspondence: * [email protected], ** [email protected]
The authors acknowledge financial support from the MIT Energy Initiative, the MIT Deshpande Center for Technological Innovation and ENN Group. Q.P., J.M. and X.H. thank the Peking University startup funding, the National Natural Science Foundation of China (grant no. 22075002), and the National Postdoctoral Programme for Innovative Talents (grant no. BX2021002). L.M. acknowledges financial support from The National Key Research and Development Program of China (grant no.2020YFA0715000). B.N. and S.G. acknowledge funding from the Office of the Executive Vice President for Research and Innovation at the University of Louisville. This research used resources of the National Energy Research Scientific Computing Center, a U.S. Department of Energy (DOE_ Office of Science User Facility supported by the Office of Science of the U.S. DOE under Contract No. DE-AC02-05CH11231. This research used resources of the Advanced Photon Source, a U.S. DOE Office of Science user facility operated for the DOE Office of Science by Argonne National Laboratory under Contract No. DE-AC02-06CH11357.
The U.S. Department of Energy's APS at Argonne National Laboratory is one of the world’s most productive x-ray light source facilities. Each year, the APS provides high-brightness x-ray beams to a diverse community of more than 5,000 researchers in materials science, chemistry, condensed matter physics, the life and environmental sciences, and applied research. Researchers using the APS produce over 2,000 publications each year detailing impactful discoveries and solve more vital biological protein structures than users of any other x-ray light source research facility. APS x-rays are ideally suited for explorations of materials and biological structures; elemental distribution; chemical, magnetic, electronic states; and a wide range of technologically important engineering systems from batteries to fuel injector sprays, all of which are the foundations of our nation’s economic, technological, and physical well-being.
Argonne National Laboratory seeks solutions to pressing national problems in science and technology. The nation's first national laboratory, Argonne conducts leading-edge basic and applied scientific research in virtually every scientific discipline. Argonne researchers work closely with researchers from hundreds of companies, universities, and federal, state and municipal agencies to help them solve their specific problems, advance America's scientific leadership and prepare the nation for a better future. With employees from more than 60 nations, Argonne is managed by UChicago Argonne, LLC, for the U.S. DOE Office of Science.
The U.S. Department of Energy's Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information, visit the Office of Science website.
