Abdominal aortic aneurysms (AAA) cause approximately 10,000 deaths each year in the United States. An AAA occurs when a section of the aorta that runs through the abdomen is weakened and begins to bulge. If the aneurysm ruptures, a person has a 90% chance of dying. As of now, the only treatment for an AAA is surgery and that only works if it is caught in time. A new study, published in the journal ACS Nano, involved experiments performed at the U.S. Department of Energy’s Advanced Photon Source (APS), which revealed a way to use nanofibers to target AAAs. These nanofibers may one day be deployed to diagnose AAAs earlier and to deliver targeted drugs to treat AAAs, reducing the number of people who die from this condition.
At about an inch in diameter and a foot long, the aorta is the largest artery in the body, and it supplies blood to most of the major organs. An abdominal aortic aneurysm (AAA) is a weakened section of the aorta that begins to balloon and can eventually rupture, putting the person at severe risk of death. AAAs develop over a prolonged period of time so tools that make it easier to identify them—and to direct pharmaceuticals to the affected area—could save lives. This new study reports an exciting advancement in this direction—the development of a nanofiber tracer that localizes to AAAs.
The researchers had previous success developing targeted nanotherapeutics for other conditions using peptide amphiphiles. Peptide amphiphiles are molecules that contain a targeting peptide (a short protein sequence designed to interact with a specific protein or enzyme in the body) connected to a hydrophobic tail. When the peptide amphiphiles are placed in liquid, the tails collect together, assembling into a nanofiber structure that leaves the targeting peptides exposed. These peptide amphiphiles can also be mixed with fluorescently tagged amphiphiles so that the assembled nanofibers can be visualized with a fluorescent microscope.
While the causes of AAAs are not completely understood, they are known to involve the breakdown of a key protein in the aorta, called elastin, by a family of enzymes called matrix metalloproteinases (MMP). These researchers took advantage of this information when designing their tracers by creating peptide amphiphiles that specifically target either fragmented elastin, matrix metalloproteinase 2 (MMP-2), or membrane type 1 matrix metalloproteinase (MT1-MMP).
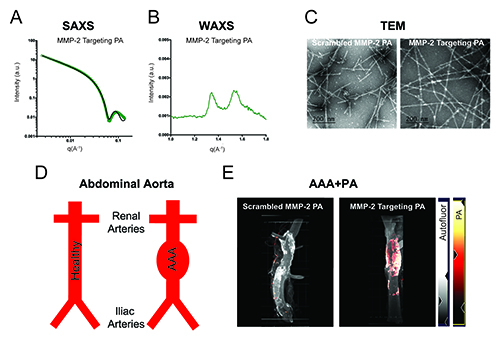
After designing and synthesizing the peptide amphiphiles, the researchers carried out a series of experiments employing a variety of techniques (Fig. 1). They used transmission electron microscopy to confirm that the peptide amphiphiles were successfully forming nanofibers. They then performed x-ray scattering experiments at beamline 5-ID-D of the DuPont-Northwestern-Dow Collaborative Access Team (DND-CAT) at the APS to get more detailed information about the molecular structures that makeup the nanofibers. The APS is an Office of Science user facility at Argonne National Laboratory.
Next, the researchers tested the ability of the three peptide amphiphiles to target AAAs, using a rat model of AAA that mimics the key features of human AAA. The researchers employed a special type of microscopy called light sheet fluorescence microscopy to visualize the fluorescently labeled amphiphiles within the nanofibers. They found that each of the three peptide amphiphiles successfully localized to the site of the aneurysm with the MMP-2 targeting peptide amphiphile showing the strongest targeting.
While the MMP-2 targeting peptide amphiphile predominantly localized to the aorta of the rats, it was also found in the kidney, liver, spleen, and lung, although at much lower levels. The tracer remained localized to the AAA site for at least 24 hours but was largely cleared by 72 hours.
Together, these findings suggest that MMP-2 targeting peptide amphiphile nanofibers could one day be used as a diagnostic tool to detect early phases of AAA development. Additionally, these nanofibers could be harnessed for a targeted drug delivery system for a nanotherapeutic that could deliver a higher effective dose at the site of the aneurysm and have fewer side effects than drugs delivered to the entire body, transforming treatment options for this deadly condition. ― Neil Savage
See: Benjamin T. Ledford1,2, Adam W. Akerman2, Kui Sun2, David C. Gillis1,2, Jenna M. Weiss2, Johnny Vang2, Smaranda Willcox2, Tristan D. Clemons3, Hiroaki Sai3, Ruomeng Qiu3, Mark R. Karver3, Jack D. Griffith2, Nick D. Tsihlis1,2, Samuel I. Stupp3, John S. Ikonomidis2, and Melina R. Kibbe1,2*, “Peptide Amphiphile Supramolecular Nanofibers Designed to Target Abdominal Aortic Aneurysms,” ACS Nano 16, 7309 (2022). DOI: 10.1021/acsnano.1c06258
Author affiliations: 1University of Virginia, 2University of North Carolina at Chapel Hill, 3Northwestern University
Correspondence: * [email protected]
This study was supported, in part, by funding from the University of North Carolina School of Medicine. T.D.C. acknowledges funding support from an American Australian Association Fellowship and the Center for Regenerative Nanomedicine at the Simpson Querrey Institute for BioNanotechnology at Northwestern University. Technical support was provided by several core facilities at UNC Chapel Hill including: the UNC Microscopy Services Lab, the Small Animal Imaging Facility at the Biomedical Research Imaging Center, the R. L. Juliano Bioinformatics Core facility, and the Neuroscience Microscopy Core. The TEM studies were supported by a grant to J.D.G. from the National Institutes of Health (5R0)-ES031635. The Microscopy Services Laboratory, Department of Pathology and Laboratory Medicine, is supported in part by P30 CA016086 Cancer Center Core Support Grant to the UNC Lineberger Comprehensive Cancer Center. Research reported in this publication was supported in part by the North Carolina Biotech Center Institutional Support Grant 2016-IDG-1016. MST and CD spectroscopy experiments were conducted at the UNC Macromolecular Interactions Facility, which is supported by the National Cancer Institute of the National Institutes of Health under award number P30CA016086. DND-CAT is supported by Northwestern University, E.I. DuPont de Nemours & Co., and The Dow Chemical Company. Peptide amphiphile synthesis was performed in the Peptide Synthesis Core Facility of the Simpson Querrey Institute at Northwestern University. The U.S. Army Research Office, the U.S. Army Medical Research and Materiel Command, and Northwestern University provided funding to develop this facility, and ongoing support is being received from the Soft and Hybrid Nanotechnology Experimental (SHyNE) (NSF ECCS-2025633). This research used resources of the Advanced Photon Source, a U.S. Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under Contract No. DE-AC02-06CH11357.
The U.S. Department of Energy's APS at Argonne National Laboratory is one of the world’s most productive x-ray light source facilities. Each year, the APS provides high-brightness x-ray beams to a diverse community of more than 5,000 researchers in materials science, chemistry, condensed matter physics, the life and environmental sciences, and applied research. Researchers using the APS produce over 2,000 publications each year detailing impactful discoveries, and solve more vital biological protein structures than users of any other x-ray light source research facility. APS x-rays are ideally suited for explorations of materials and biological structures; elemental distribution; chemical, magnetic, electronic states; and a wide range of technologically important engineering systems from batteries to fuel injector sprays, all of which are the foundations of our nation’s economic, technological, and physical well-being.
Argonne National Laboratory seeks solutions to pressing national problems in science and technology. The nation's first national laboratory, Argonne conducts leading-edge basic and applied scientific research in virtually every scientific discipline. Argonne researchers work closely with researchers from hundreds of companies, universities, and federal, state and municipal agencies to help them solve their specific problems, advance America's scientific leadership and prepare the nation for a better future. With employees from more than 60 nations, Argonne is managed by UChicago Argonne, LLC, for the U.S. DOE Office of Science.
The U.S. Department of Energy's Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information, visit the Office of Science website.
