Astronomers have discovered thousands of planets orbiting other stars. Many of these planets are rocky like Earth, but considerably bigger. Researchers wonder if these “Super Earths” could support life. One of the requirements for life as we know it is plate tectonics, the slow churning of a planet’s interior that renews the surface, regulates the carbon cycle, and keeps liquid water available. Now, researchers have used the U.S. Department of Energy's Advanced Photon Source (APS) to understand how common silicate minerals would behave under the incredible pressures deep in the mantle of a Super Earth. Their findings, published in the Proceedings of the National Academy of Sciences of the United States of America, reveal a new phase of mineral never before reported.
Magnesium silicates are common minerals that comprise the bulk of rocky planets. Geophysicists have a fairly good understanding of how magnesium (and iron) silicates behave inside of the Earth: They form a mineral phase called bridgmanite in the Earth's deep mantle and this defines a major structural boundary in our planet. But under the extraordinarily high pressures in the mantle of a Super Earth, researchers suspect magnesium and iron silicates would form a mineral phase with a structure similar to that of thorium phosphide. Unfortunately, we have no way to reproduce those pressures in the lab.
However, geophysicists can study magnesium germanates instead. Germanium is chemically akin to silicon, sitting directly beneath it in the periodic table. But its larger size makes it more easily compressible, and magnesium germanates pass through the same phase transitions as magnesium silicates do, at lower pressures.
A team of researchers from the Carnegie Institution for Science, Princeton University, University of Nevada Las Vegas, and The University of Chicago used a laser-heated diamond anvil to compress magnesium germinate (Mg2GeO4) to pressures as high as 275 gigapascals (GPa). X-ray beamlines at the APS operated by the GeoSoilEnviroCARS collaborative access team (beamline 13-ID-D) and the Argonne X-ray Science Division's HPCAT-XSD (beamline 16-ID-B) were both used for various parts of the experiment. This was the first time x-ray diffraction was performed in situ at such high pressures on a germanate, and it was done in remote access mode using NoMachine’s NX software at HPCAT-XSD.
The x-ray diffraction results showed that at first, magnesium germanate passed through the same phases geophysicists see in magnesium silicates: First, a phase in which each germanium atom is surrounded by four oxygen atoms, and then at higher pressures, it changed to a phase in which each germanium atom is surrounded by six oxygens. And then, as the pressure climbed even higher, the x-ray diffraction revealed something surprising: Each germanium atom was coordinated with eight oxygen atoms! The researchers believe this is the first time anyone has ever reported an 8-coordinate silicate or germanate.
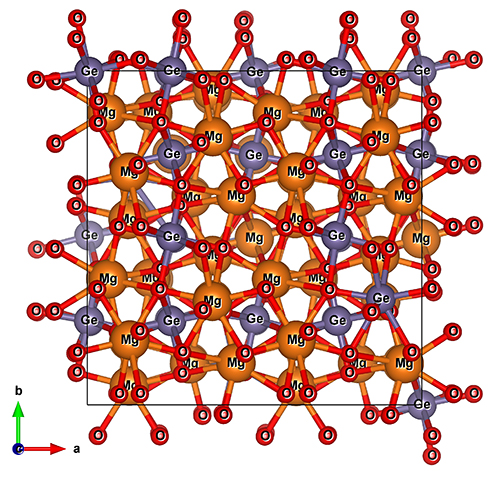
There was another surprise, too. The structure of the material became somewhat disordered, with magnesium atoms swapping position interchangeably with germanium atoms (Fig. 1).
The authors also performed theoretical computations based on density functional theory. The computations show that the completely ordered structure gradually disorders with increasing temperature resulting in a partially disordered tetragonal structure, experimentally indistinguishable from the completely disordered thorium phosphide structure.
In 6-coordinated minerals, magnesium is 25% larger than germanium. But it is possible that the extremely high pressures compact the magnesium and germanium atoms so much that they become close to the same size and able to swap in and out of each other’s position in the crystal matrix. The unusual 8-coordinated atomic structure causes a very significant increase in density of the mineral of about 2.4%, which may affect the behavior of the deep mantle: compression and shear wave propagation in material is dependent on material density. And the disordered nature of the crystal may lead to enhanced miscibility of chemical components and impurities at these high pressures. Both higher density and higher miscibility could lead to a planet with very different internal characteristics and plate tectonics than the Earth we know. It’s also possible that other oxide minerals adopt a similar 8-coordinate structure at Super Earth pressures.
The next step in this research is to add iron to the magnesium germinates and run the same experiments, checking to see whether the presence of iron changes the mineral structure.
― Kim Krieger
See: Rajkrishna Dutta1, 2, Sally June Tracy1, R. E. Cohen1, Francesca Miozzi1, Kai Luo1, Jing Yang1, Pamela C. Burnley3,Dean Smith4, Yue Meng4, Stella Chariton5, Vitali B. Prakapenka5, and Thomas S. Duffy2, “Ultrahigh-pressure disordered eight-coordinated phase of Mg2GeO4: Analogue for super-Earth mantles,” Proc. Natl. Acad. Sci. U.S.A. 119 (8), e2114424119 (February 14, 2022). DOI: 10.1073/pnas.2114424119
Author affiliations: 1Carnegie Institution for Science, 2Princeton University, 3University of Nevada, 4Argonne National Laboratory, 5The University of Chicago
Correspondence: * [email protected]
This research was supported by the National Science Foundation (NSF)—Earth Sciences (EAR-1836852). R.D. is grateful for support from the Carnegie Endowment. GeoSoilEnviroCARS is supported by the NSF—Earth Sciences (EAR-1634415) and the U.S. Department of Energy (DOE) Geosciences (DE-FG02-94ER14466). HPCAT-XSD operations are supported by DOE National Nuclear Security Administration Office of Experimental Sciences. R.E.C. gratefully acknowledges the Gauss Centre for Supercomputing e.V. (https://www.gauss-centre.eu/) for funding this project by providing computing time on the GCS SupercomputerSuperMUC-NG at Leibniz Supercomputing Centre (LRZ,http://www.lrz.de). This research used resources of the Advanced Photon Source, a U.S. DOE Office of Science user facility, operated for the DOE Office of Science by Argonne National Laboratory under contract no. DE-AC02-06CH11357.
The Carnegie Institution for Science press release on this subject can be read here.
The U.S. Department of Energy's APS at Argonne National Laboratory is one of the world’s most productive x-ray light source facilities. Each year, the APS provides high-brightness x-ray beams to a diverse community of more than 5,000 researchers in materials science, chemistry, condensed matter physics, the life and environmental sciences, and applied research. Researchers using the APS produce over 2,000 publications each year detailing impactful discoveries, and solve more vital biological protein structures than users of any other x-ray light source research facility. APS x-rays are ideally suited for explorations of materials and biological structures; elemental distribution; chemical, magnetic, electronic states; and a wide range of technologically important engineering systems from batteries to fuel injector sprays, all of which are the foundations of our nation’s economic, technological, and physical well-being.
Argonne National Laboratory seeks solutions to pressing national problems in science and technology. The nation's first national laboratory, Argonne conducts leading-edge basic and applied scientific research in virtually every scientific discipline. Argonne researchers work closely with researchers from hundreds of companies, universities, and federal, state and municipal agencies to help them solve their specific problems, advance America's scientific leadership and prepare the nation for a better future. With employees from more than 60 nations, Argonne is managed by UChicago Argonne, LLC, for the U.S. DOE Office of Science.
The U.S. Department of Energy's Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information, visit the Office of Science website.
