The original Pacific Northwest National Laboratory press release by Susan Bauer can be read here.
Millions of medical imaging procedures each year rely on radioactive technetium. One of its radioisotopes decays quickly and is useful as a tracer material in nuclear medicine. But another, technetium-99, is very long-lived, poses a risk to the environment, and is a potential health threat. Technetium can be found in nuclear reactor waste and at sites that processed uranium for nuclear weapons during the Cold War, such as the Hanford Site in Washington State. The most prevalent form of technetium—called pertechnetate—is water soluble and has a high potential for spreading through the soil and groundwater. But now, researchers at Pacific Northwest National Laboratory (PNNL), Florida International University, and the Illinois Institute of Technology are closer to understanding how high concentrations of technetium-99 can be treated by simple iron, which is inexpensive and readily available. Their results, based in large part upon research at the U.S. Department of Energy’s Advanced Photon Source (APS), were published in the journal Communications Chemistry.
The team recently completed experiments in which almost all—99.8%—of the technetium was removed from a liquid solution after one month in contact with iron particles. This occurs as the iron oxidizes or loses electrons in a chemical process called reductive removal.
“We just let them sit in contact with each other,” said Daria Boglaienko, an environmental scientist at PNNL. “That’s what’s different about this study. We did not control any parameters such as acidity or temperature or oxygen content. We just let the reaction unfold spontaneously, as it would in nature if these two compounds came in contact with each other.”
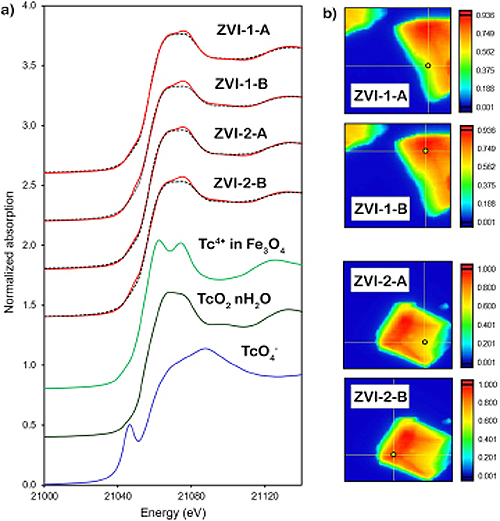
The team went on to analyze the reaction products at the atomic scale with sophisticated instruments at PNNL’s Radiochemical Processing Laboratory and two U.S. Department of Energy (DOE) Office of Science user facilities—EMSL, the Environmental Molecular Sciences Laboratory at PNNL, and the APS, an Office of Science user facility at Argonne National Laboratory. Work at the APS included x-ray absorption near edge structure and extended x-ray absorption fine structure measurements of the technetium K-edge in granular zero valent iron (ZVI) carried out at the X-ray Science Division Spectroscopy Group’s 20-ID-C x-ray beamline (Fig. 1). In addition, 2-D elemental maps were collected based on x-ray fluorescence mapping.
Previous studies observed this reaction under controlled anaerobic conditions and/or with relatively low loadings of technetium. But this study is the first to show how the reaction occurs spontaneously under aerobic conditions with high concentrations of technetium, and to understand how the technetium is incorporated into the iron mineral lattice.
The research revealed that clusters of technetium partially incorporate into iron minerals, such as magnetite, during simultaneous iron oxidation and mineralization. This is the same incorporation mechanism that would be expected in a natural system.
“The fact that this experiment was conducted in conditions that would spontaneously occur with the introduction of iron, known as zero valent iron, has positive implications for environmental cleanup efforts in the subsurface or nuclear waste treatment systems,” said corresponding author Tatiana Levitskaia, a chemist at PNNL.
The researchers also found that the presence of technetium slows the oxidation of the metallic iron and its ultimate transformation from ferrihydrite to magnetite. The slower process gives the technetium time to incorporate into the magnetite.
Once lodged within the crystalline structure, the technetium does not re-oxidize back to its more mobile form of pertechnetate—it is instead sequestered for the long term, which minimizes the risk of release back into the groundwater or subsurface environment.
The research team continues to work with zero valent iron. They have tested its ability to separate and sequester technetium at Hanford, where cleanup will involve vitrification—or turning liquid wastes into glass—for safe long-term storage. A portion of the technetium may not fully incorporate into the glass and end up in a secondary waste stream. Zero valent iron may be effective in removing technetium from these secondary wastes.
The research was supported by DOE’s Office of Environmental Management and its Minority Serving Institution Partnership Program. It was performed as part of DOE’s Technetium Management Project.
Daria Boglaienko1, Jennifer A. Soltis1, Ravi K. Kukkadapu1, Yingge Du1, Lucas E. Sweet1, Vanessa E. Holfeltz1, Gabriel B. Hall1, Edgar C. Buck1, Carlo U. Segre2, Hilary P. Emerson1, Yelena Katsenovich3, and Tatiana G. Levitskaia1*, “Spontaneous redox continuum reveals sequestered technetium clusters and retarded mineral transformation of iron,” Commun. Chem. 3, 87 (2020). DOI: 10.1038/s42004-020-0334-x. ©2020 Springer Nature Limited
Author affiliations: 1Pacific Northwest National Laboratory, 2Illinois Institute of Technology, 3Florida International University
Correspondence: *[email protected]
Home page illustration: https://pixy.org/2346367/
This research was supported by the U.S. Department of Energy (DOE) Office of Environmental Management and performed as part of the Technetium Management Hanford Site project at PNNL operated by Battelle for the U.S. DOE under Contract No. DE-AC05–76RL01830. This work was in part supported by the DOE Minority Serving Institution Partnership Program (MSIPP) managed by the Savannah River National Laboratory under SRNS contract DE-AC09-08SR22470. We also acknowledge ‘2019 APS/IIT XAFS Summer School’ in Chicago, IL. This research used resources of the Advanced Photon Source, an Office of Science User Facility operated for the U.S. DOE Office of Science by Argonne National Laboratory, and was supported by the U.S. DOE under Contract No. DE-AC02-06CH11357, and the Canadian Light Source and its funding partners.
The U.S. Department of Energy's APS is one of the world’s most productive x-ray light source facilities. Each year, the APS provides high-brightness x-ray beams to a diverse community of more than 5,000 researchers in materials science, chemistry, condensed matter physics, the life and environmental sciences, and applied research. Researchers using the APS produce over 2,000 publications each year detailing impactful discoveries, and solve more vital biological protein structures than users of any other x-ray light source research facility. APS x-rays are ideally suited for explorations of materials and biological structures; elemental distribution; chemical, magnetic, electronic states; and a wide range of technologically important engineering systems from batteries to fuel injector sprays, all of which are the foundations of our nation’s economic, technological, and physical well-being.
Pacific Northwest National Laboratory advances the frontiers of knowledge, taking on some of the world’s greatest science and technology challenges. Distinctive strengths in chemistry, Earth sciences, and data analytics are the heart of our science mission, laying a foundation for innovations that improve America’s energy resiliency and enhance our national security.
Argonne National Laboratory seeks solutions to pressing national problems in science and technology. The nation's first national laboratory, Argonne conducts leading-edge basic and applied scientific research in virtually every scientific discipline. Argonne researchers work closely with researchers from hundreds of companies, universities, and federal, state and municipal agencies to help them solve their specific problems, advance America's scientific leadership and prepare the nation for a better future. With employees from more than 60 nations, Argonne is managed by UChicago Argonne, LLC, for the U.S. DOE Office of Science.
The U.S. Department of Energy's Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information, visit the Office of Science website.
