For centuries, microscopy has been an invaluable tool for understanding nature. Fluorescence microscopy in particular can image a diverse array of molecules. For example, a molecule can be conjugated to a special protein that emits green light. Such conjugation allows researchers to identify a molecule in a cell or tissue by finding regions that fluoresce green. While this technique is incredibly powerful, it is challenging to obtain an optical resolution better than 200 nanometers. In recent work carried out at two U.S. Department of Energy (DOE) x-ray light sources, including the Advanced Photon Source (APS), a novel tool was developed to obtain impressive spatial resolutions (e.g., 36 nanometers) in intact cells. This was accomplished by adapting lanthanide-binding tags in conjunction with x-ray fluorescence microscopy. Since lanthanide-binding tags offer many unique advantages, this novel work may allow for previously inaccessible structures and molecules to be imaged at a high resolution.
Many key discoveries have been made by knowing the location and activity of a given molecule or a group of molecules. Highlighting how important fluorescence microscopy is in science, the 2014 Nobel Prize in Chemistry was awarded to three individuals for advancements in fluorescence microscopy that allow for super-resolution imaging. Prior to the advent of super-resolution microscopy, light microscopy could not reliably obtain a resolution greater than 200 nanometers or distinguish objects less than 200 nanometers apart. This is problematic because many objects, such as many viruses, are smaller than 200 nanometers.
One unique form of fluorescence microscopy is x-ray fluorescence microscopy, which utilizes x-ray beams to perform imaging. Recent technological advances have made x-ray fluorescence microscopy quite powerful and this technique has been used to study important biological processes, such as fertilization, Alzheimer’s disease, Parkinson’s disease, chemotherapy, and malaria. However, this technique has historically not been capable of super-resolution and has been limited to ~300 nanometers laterally (two dimensions) and ~1,000 nanometers axially (three dimensions). Unlike traditional fluorescence microscopy, there is also a dearth of x-ray sensitive tags that can be paired with molecules to image them.
Recent work by researchers from Brookhaven National Laboratory, Boston University, Argonne National Laboratory, Oregon Health Science University, and MIT directly addressed these challenges. The resolution obstacle discussed above was solved by using advanced optics including high-resolution zone plates and the multilayer Laue x-ray lenses, which allows for spatial resolutions approaching 10 nanometers, a ~20-fold improvement over the conventional diffraction resolution limit. The latter tagging problem was addressed by customizing short peptides (15-20 amino acids long) referred to as lanthanide-binding tags.
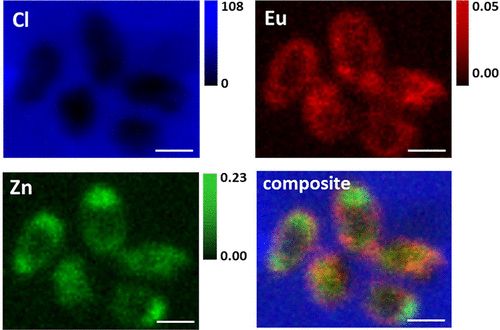
The metallic chemical elements (rare-earth elements) with atomic numbers (proton numbers) 57-71 make up the lanthanide series. Other techniques have previously used lanthanide binding tags for important research techniques such as nuclear magnetic resonance spectroscopy, magnetic resonance imaging, photoluminescence, and x-ray crystallography. Creatively, these researchers have adapted lanthanide-binding tags for x-ray fluorescence microscopy. These tags are well-suited for biological imaging because lanthanide elements are extremely rare within living organisms. The authors present a variety of beautiful, colored images in bacteria to demonstrate that these tags can be used for two- and three-dimensional imaging. Figure 1 shows high-resolution images from the APS Bionanoprobe of elements.
The Bionanoprobe, which was at the Life Sciences Collaborative Access Team beamline 21-ID-D and now is at the X-ray Science Division (XSD) Microscopy Group's beamline 9-ID-B, both at the DOE’s APS, was used to perform two-dimensional x-ray fluorescence microscopy. The Hard X-ray Nanoprobe beamline 3-ID at the DOE’s National Synchrotron Light Source II (NSLS-II) at Brookhaven National Laboratory was used for the three-dimensional x-ray nanotomography work. Both the APS and NSLS-II are Office of Science user facilities.
This research demonstrates that x-ray imaging can be performed in the X, Y, and Z dimensions using lanthanide-binding tags. Since these tags have an incredibly high affinity for lanthanides, they can be used at low, non-toxic concentrations. Protein sequences are amenable to their incorporation and their small size means that they don’t hinder a protein’s ability to carry out its physiological functions. These data demonstrate that this approach can be used to generate high-quality images with a nanoscale resolution. Since x-rays have a long penetration depth, this method could be feasibly applied to imaging in tissues. Future research should build upon these findings and aim to explore what novel insights can be gleaned from the unique combination of lanthanide-binding tags and x-ray imaging. ― Alicia Surrao
See: Tiffany W. Victor1, Katherine H. O’Toole2, Lindsey M. Easthon2, Mingyuan Ge1, Randy J. Smith1, Xiaojing Huang1, Hanfei Yan1, Yong S. Chu1, Si Chen3, Doga Gursoy3, Martina Ralle4, Barbara Imperiali5, Karen N. Allen2, and Lisa M. Miller1*, “Lanthanide-Binding Tags for 3D X‑ray Imaging of Proteins in Cells at Nanoscale Resolution,” J. Am. Chem. Soc. 142, 2145 (2020). DOI: 10.1021/jacs.9b11571
Author affiliations: 1Brookhaven National Laboratory, 2Boston University, 3Argonne National Laboratory, 4Oregon Health Science University, 5Massachusetts Institute of Technology
Correspondence: *[email protected]
This work was supported by the U.S. Department of Energy (DOE) Office of Biological and Environmental Research as part of the “Environment Sensing and Response” Scientific Focus Area of the BER Genomic Science Program. T.W.V. was partially supported by the National Institutes of Health T32 Grant 5T32GM092714 and a Director’s Postdoctoral Fellowship at NSLS-II. B.I. and K.N.A. were supported by National Science Foundation MCB-1615252 and MCB-1614608, respectively. The Life Sciences Collaborative Access Team is supported by the Michigan Economic Development Corporation and the Michigan Technology Tri-Corridor (Grant 085P1000817). This research used beamline 3-ID (HXN) at the National Synchrotron Light Source II, a U.S. DOE Office of Science User Facility operated for the DOE Office of Science by Brookhaven National Laboratory under Contract No. DE-SC0012704. This research used resources of the Advanced Photon Source, a U.S. DOE Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under Contract No. DE-AC02-06CH11357.
The U.S. Department of Energy's APS is one of the world’s most productive x-ray light source facilities. Each year, the APS provides high-brightness x-ray beams to a diverse community of more than 5,000 researchers in materials science, chemistry, condensed matter physics, the life and environmental sciences, and applied research. Researchers using the APS produce over 2,000 publications each year detailing impactful discoveries, and solve more vital biological protein structures than users of any other x-ray light source research facility. APS x-rays are ideally suited for explorations of materials and biological structures; elemental distribution; chemical, magnetic, electronic states; and a wide range of technologically important engineering systems from batteries to fuel injector sprays, all of which are the foundations of our nation’s economic, technological, and physical well-being.
Argonne National Laboratory seeks solutions to pressing national problems in science and technology. The nation's first national laboratory, Argonne conducts leading-edge basic and applied scientific research in virtually every scientific discipline. Argonne researchers work closely with researchers from hundreds of companies, universities, and federal, state and municipal agencies to help them solve their specific problems, advance America's scientific leadership and prepare the nation for a better future. With employees from more than 60 nations, Argonne is managed by UChicago Argonne, LLC, for the U.S. DOE Office of Science.
The U.S. Department of Energy's Office of Science is the single largest supporter of basic research in the physical science
