 Electrochemical carbon dioxide reduction (CDR) provides an efficient green strategy for solving energy and environmental problems. It uses the electricity generated by renewable energy sources to drive the electrochemical immobilization of carbon dioxide (CO2) and convert it into high value-added chemical products. However, electrochemical CDR currently faces many challenges, including slow reaction kinetics, low selectivity of target products, and competitive hydrogen evolution reactions. Therefore, the development of a cost-effective, stable and highly efficient CDR electrocatalyst is the key to the large-scale application of electrochemical CDR technology. Using a simple solvothermal method, a multi-institution international research team synthesized a bismuth oxide nanotube precursor with a one-dimensional tubular structure that exhibits very good catalytic activity in electrochemical carbon dioxide reduction. In situ synchrotron radiation characterization carried at the U.S. Department of Energy’s Advanced Photon Source (APS) showed that the bismuth oxide catalyst precursor had a large number of structural defects after it was converted into a metal phase at the cathode side. The theoretical calculations show that the existence of defects is very helpful to improving CDR catalytic activity. The test results of both the H-type electrolytic cell and the flow cell confirm its excellent electrochemical performance. This work, published in Nature Communications, provides a viable guide for materials design applied to the large-scale implementation of electrochemical CO2 fixation.
Electrochemical carbon dioxide reduction (CDR) provides an efficient green strategy for solving energy and environmental problems. It uses the electricity generated by renewable energy sources to drive the electrochemical immobilization of carbon dioxide (CO2) and convert it into high value-added chemical products. However, electrochemical CDR currently faces many challenges, including slow reaction kinetics, low selectivity of target products, and competitive hydrogen evolution reactions. Therefore, the development of a cost-effective, stable and highly efficient CDR electrocatalyst is the key to the large-scale application of electrochemical CDR technology. Using a simple solvothermal method, a multi-institution international research team synthesized a bismuth oxide nanotube precursor with a one-dimensional tubular structure that exhibits very good catalytic activity in electrochemical carbon dioxide reduction. In situ synchrotron radiation characterization carried at the U.S. Department of Energy’s Advanced Photon Source (APS) showed that the bismuth oxide catalyst precursor had a large number of structural defects after it was converted into a metal phase at the cathode side. The theoretical calculations show that the existence of defects is very helpful to improving CDR catalytic activity. The test results of both the H-type electrolytic cell and the flow cell confirm its excellent electrochemical performance. This work, published in Nature Communications, provides a viable guide for materials design applied to the large-scale implementation of electrochemical CO2 fixation.
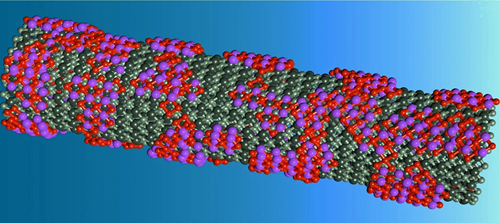
The authors first prepared a Bi2O3 nanotube (Fig. 1) with a fragmented outer surface and then converted it into a highly deficient metal NTD-Bi by a cathodic conversion method. In the H-type electrolytic cell, the Faraday efficiency of formic acid can remain between 98% and 100% with a current density achieving 36mA/cm2 during the 48-hour tests in a CO2 saturated 0.5M KHCO3 electrolyte. In the self-designed flow cell, the NTD-Bi catalytic performance of CDR to produce formate can achieve a current density up to 210 mA/cm2, and Faraday efficiency of 98%, which can last for 11-13 hours in 1M KOH electrolyte. This is the first time the technical requirements of commercial applications have been met, in which the current density should be greater than 200 mA/cm2, the productivity of formic acid should be over 95%, and the stability should reach 1000 hours. In addition, the combining of NTD-Bi with Si demonstrates the good photo-electrocatalytic performance of CDR in producing formate.
In order to explore the fine structure and chemical environment of the catalyst during the electrochemical reduction of CO2, the authors conducted synchrotron in situ and operando x-ray absorption spectroscopy (XAS) measurements at the DuPont-Northwestern-Dow Collaborative Access Team (DND-CAT) x-ray beamline 5-BM-D at the APS (the APS is an Office of Science user facility at Argonne National Laboratory). The XAS results confirmed that Bi2O3 nanotubes had been reduced to NTD-Bi before the CO2 reduction reaction (-0.24 V), in which the coordination number of Bi-Bi is 2.6±1.8, significantly smaller than the coordination number of the standard Bi metal (CN=6). Although the authors were unable to accurately determine the atomic configuration of these low coordination sites, the EXAFS results provide reliable evidence that NTD-Bi has a large number of defects or voids under actual operating conditions. The authors also used density functional theory (DFT) for simulation analysis to compare the CO2 reduction pathways on the ideal and defective Bi surfaces, which show that the presence of abundant defect sites in NTD-Bi can stabilize the *OCHO (intermediate to formate) and greatly enhance the kinetics of the reaction.
See: Qiufang Gong1, Pan Ding1, Mingquan Xu2, Xiaorong Zhu3, Maoyu Wang4, Jun Deng1, Qing Ma5, Na Han1, Yong Zhu2, Jun Lu6, Zhenxing Feng4*, Yafei Li3**, Wu Zhou2***, and Yanguang Li1****, “Structural defects on converted bismuth oxide nanotubes enable highly active electrocatalysis of carbon dioxide reduction,” Nat. Commun. 10, 2807 (2019). DOI: 10.1038/s41467-019-10819-4
Author affiliations: 1Soochow University, 2University of Chinese Academy of Sciences, 3Nanjing Normal University, 4Oregon State University, 5Northwestern University, 6Argonne National Laboratory
Correspondence: ****yanguang@suda.edu.cn, ***[email protected], **[email protected], *[email protected]
Besides multiple sources that sponsored this work, the authors especially thank the instrumental and technical support from DND-CAT, which is supported by Northwestern University, The Dow Chemical Company, and DuPont de Nemours, Inc. This research used resources of the Advanced Photon Source, a U.S. Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under Contract No. DE-AC02-06CH11357.
Argonne National Laboratory seeks solutions to pressing national problems in science and technology. The nation's first national laboratory, Argonne conducts leading-edge basic and applied scientific research in virtually every scientific discipline. Argonne researchers work closely with researchers from hundreds of companies, universities, and federal, state and municipal agencies to help them solve their specific problems, advance America's scientific leadership and prepare the nation for a better future. With employees from more than 60 nations, Argonne is managed by UChicago Argonne, LLC, for the U.S. DOE Office of Science.
The U.S. Department of Energy's Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information, visit the Office of Science website.
