 These days, when one has an epiphany, it's a figurative solid-state light bulb that lights up over one’s head. Those energy-conscious, white-light spectrum bulbs could benefit from the characteristics of semiconducting quantum dot nanoparticles – an alternative to the currently-used solid phosphors. In order to leverage these nanoparticles' valuable properties, material scientists need a thorough understanding of their lattice structure and energy dynamics and how those characteristics differ between a bulk sample of the crystal and the nano-sized one. Unfortunately, the most widely-used materials characterization methods are tailored to bulk crystals. However, recent research efforts carried out at the U.S. Department of Energy’s Advanced Photon Source (APS) and National Synchrotron Light Source II (NSLS-II) have yielded new structural and energetic insights into quantum dot nanoparticles by pairing a new technique for precisely-generating cadmium selenide quantum dots with the x-ray scattering techniques. In addition to demonstrating the advantages of this combination, the researchers concluded that the nanoparticle material could be intentionally manipulated to select for certain desired electronic behaviors.
These days, when one has an epiphany, it's a figurative solid-state light bulb that lights up over one’s head. Those energy-conscious, white-light spectrum bulbs could benefit from the characteristics of semiconducting quantum dot nanoparticles – an alternative to the currently-used solid phosphors. In order to leverage these nanoparticles' valuable properties, material scientists need a thorough understanding of their lattice structure and energy dynamics and how those characteristics differ between a bulk sample of the crystal and the nano-sized one. Unfortunately, the most widely-used materials characterization methods are tailored to bulk crystals. However, recent research efforts carried out at the U.S. Department of Energy’s Advanced Photon Source (APS) and National Synchrotron Light Source II (NSLS-II) have yielded new structural and energetic insights into quantum dot nanoparticles by pairing a new technique for precisely-generating cadmium selenide quantum dots with the x-ray scattering techniques. In addition to demonstrating the advantages of this combination, the researchers concluded that the nanoparticle material could be intentionally manipulated to select for certain desired electronic behaviors.
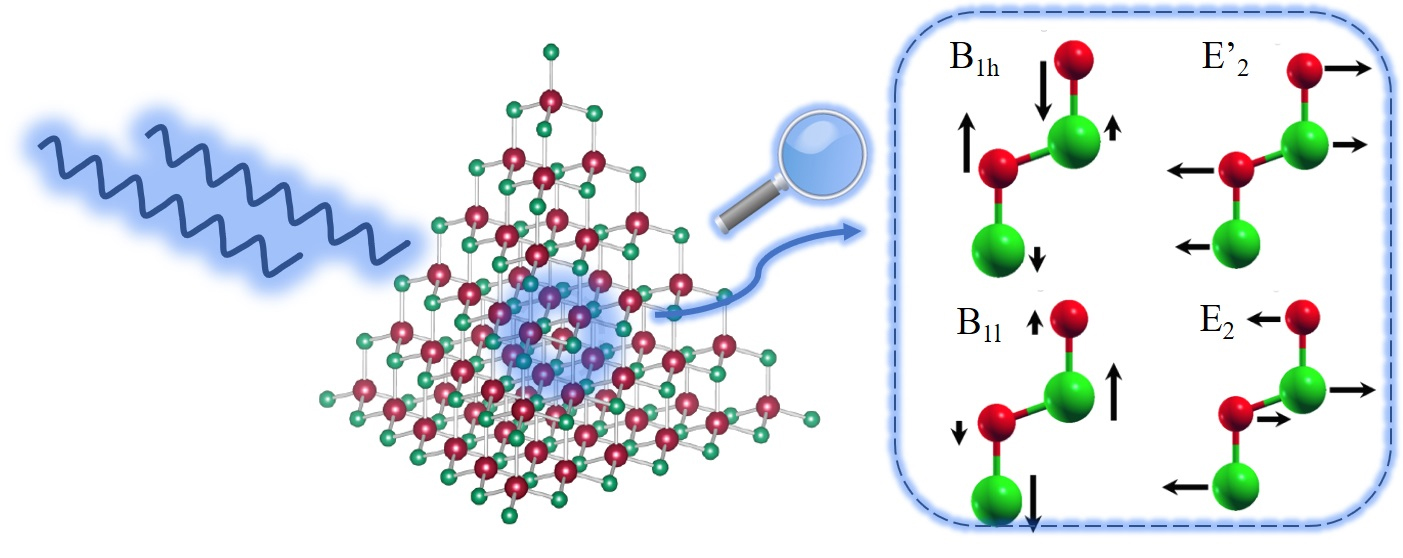
Quantum dot nanoparticles are of interest in applications ranging from electronics to biology because of their optoelectronic properties. These include their size-tunable band gaps, high efficiency at producing light, and long-term stability. The key challenges in utilizing a specific nanoparticle are two-fold: determining its structure with atomic precision, and detailing the energy dynamics of its lattice. When characterizing the lattice, scientists are most interested in atomic bonding and the behavior of atomic collective excitations (phonons). Phonons are quantum atomic vibrations generated in the lattice and from the phonon density of states, scientists can calculate many thermodynamic properties relevant in optoelectronic applications.
The research team in this study, comprising members from Columbia University, Miami University, Argonne National Laboratory, Brookhaven National Laboratory, and the American Physical Society, began by generating gram quantities of identical cadmium selenide quantum dot nanoparticles. These quantum dots are important and novel because they are atomically precise. This atomical precision allowed the team to determine the dots’ structure by applying pair distribution function analysis to powder x-ray scattering diffraction data taken of the nanoparticles at beamline 28-ID-2 at NSLS-II, an Office of Science user facility at Brookhaven National Laboratory.
The combination of atomical precision and a structure solution allowed the team to use density functional theory to derive theoretical values for the phonon density of states, which they then measured using the X-ray Science Division HERIX spectrometer at Sector 30, beamline 30-D-B,C, of the APS, an Office of Science user facility at Argonne. The team subjected bulk and nanoparticle samples (of three different sizes) to applied high-energy resolution inelastic x-ray scattering with an overall energy resolution of 1.5 milli-electronvolt. Figure 1 shows a depiction of this scattering and the vibrational states in the nanoparticle the team was hoping to find. They reduced this scattering data and from it calculated measured values for the phonon density of states for all samples.
When the team compared the phonon density of states – both the theoretical values and the measured ones – for the nanoparticles and the bulk sample, they found that the peaks for the nanoparticles were blue-shifted with respect to the bulk sample. They also found that the peaks which appeared in the bulk sample were significantly broader and less distinct in the nanoparticles' phonon density of states.
Because of the small size of the nanoparticles, the team had expected their features in the phonon density of states to be sharper than the bulk sample rather than flattened. Since each sample was atomically precise, they concluded that this flattening occurred because the lengths of the atomic bonds varied across an individual quantum dot sample.
The team was surprised to discover the theoretical calculations predicted a red-shift rather than the blue-shift that was observed. The team found that by adding heavy surface ligands to the calculations, they could replicate the blue-shift observed in their measurements. The team concluded that it should be possible to vary the surface ligand's mass to manipulate the phonon density of states, and thus manipulate the phonon frequencies, which is a result worthy of a very bright light bulb indeed.
- Mary Alexandra Agner
See: Chenyang Shi1*, Alexander N. Beecher1, Yan Li2, Jonathan S. Owen1, Bogdan M. Leu3,4, Ayman H. Said3, Michael Y. Hu3, and Simon J.L. Billinge1,5**, “Size-Dependent Lattice Dynamics of Atomically Precise Cadmium Selenide Quantum Dots,” Phys. Rev. Lett. 122, 026101 (2019). DOI: 10.1103/PhysRevLett.122.026101
Author affiliations: 1Columbia University, 2American Physical Society, 3Argonne National Laboratory, 4Miami University, 5Brookhaven National Laboratory
Correspondence: *[email protected], **[email protected]
Work in the Billinge group was supported by the U.S. Department of Energy (DOE) Office of Science-Basic Energy Sciences under Contract No. DE-SC0012704. Work on the nanoparticle synthesis and characterization was supported by the National Science Foundation through Contract No. NSF-CHF-1151172. The National Synchrotron Light Source II is a U.S. DOE Office of Science User Facility operated for the DOE Office of Science by Brookhaven National Laboratory under Contract No. DE-SC0012704. Computations made use of resources of the National Energy Research Scientific Computing Center, a DOE Office of Science User Facility supported by the Office of Science of the U.S. Department of Energy under Contract No. DE-AC02-05CH11231. This research used resources of the Advanced Photon Source, a U.S. DOE Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under Contract No. DE-AC02-06CH11357.
Argonne National Laboratory seeks solutions to pressing national problems in science and technology. The nation's first national laboratory, Argonne conducts leading-edge basic and applied scientific research in virtually every scientific discipline. Argonne researchers work closely with researchers from hundreds of companies, universities, and federal, state and municipal agencies to help them solve their specific problems, advance America's scientific leadership and prepare the nation for a better future. With employees from more than 60 nations, Argonne is managed by UChicago Argonne, LLC for the U.S. Department of Energy's Office of Science.
The U.S. Department of Energy's Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information, visit the Office of Science website.
