The original University of Michigan press release by Jim Erickson can be read here.
A new study by University of Michigan (U-M) and Purdue University researchers using the U.S. Department of Energy’s Advanced Photon Source (APS) has revealed the molecular structure of a protein produced by the Zika virus that is thought to be involved in the virus's reproduction and its interaction with a host's immune system.
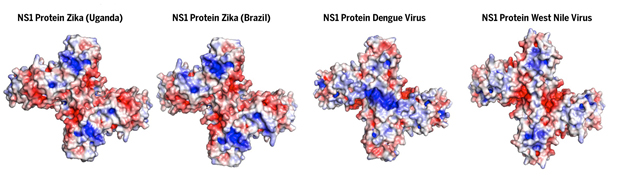
The results provide scientists around the globe with new information about the NS1 protein's role in Zika virus infections, and expands scientists' understanding of the flavivirus family, which also includes dengue, West Nile and yellow fever.
“Having the structure of the full-length Zika NS1 provides new information that could help guide the design of a potential vaccine or antiviral drugs,” said senior author Janet Smith, director of the Center for Structural Biology (CSB) at the U-M Life Sciences Institute (LSI), where her lab is located, and professor of biological chemistry at the U-M Medical School.
“Researchers are still working to understand precisely how Zika and other flaviviruses interact with an infected person’s immune system,” she said. “Having these atomic-level details can help scientists to ask better questions and to design more thoughtful experiments as we continue to learn new information.”
The findings were published in Nature Structural & Molecular Biology. Earlier this year, scientists in China published a partial structure.
The mosquito-borne Zika virus has been around for decades, but recently grew into an international health emergency following its association with severe birth defects and with Guillain-Barré syndrome, and its rapid spread in Central and South America.
There is currently no treatment or vaccine, though several companies have announced plans to try to develop one.
“Despite its similarity to other related viruses, we found the Zika NS1 structure had a few important differences,” said W. Clay Brown, scientific director of the CSB’s high-throughput protein lab and co-first author of the study.
The new 3-D structure, which was obtained using x-ray crystallography and electron microscopy, revealed that the outer surface of the Zika NS1 protein has substantially different electrical-charge properties than those of other flaviviruses—indicating it may interact differently with the members of an infected person’s immune system.
This study was also the first to capture the molecular structure of flexible loops on the wing domains of the protein, which had been hidden from view in previous studies.
"From NS1 structure studies in dengue, it was thought this loop flipped up, but our study in Zika virus shows it flips down from the wings," said co-author Richard Kuhn, professor of biological sciences at Purdue and director of the Purdue Institute of Inflammation, Immunology and Infectious Disease. "This is very important because it indicates an interaction with the cellular membrane of the host and a possible mechanism by which NS1 carries out its multiple functions.
"Seeing this difference provides new insights that help us better understand the NS1 protein," said Kuhn, who was a member of the research team that first determined the structure of the Zika virus. "Understanding its structure and functions help us identify targets for inhibitors to block important viral processes and treat infection."
The team also looked at changes in the genetic sequence of the Zika NS1 protein over time, noted David L. Akey, a research scientist in Smith's lab and the study's other lead author.
"Just as the common cold and flu viruses change over time, Zika virus changed during its spread around the world so that NS1 in Brazilian infections looks different to the immune system than its African ancestor," Akey said.
The NS1 (nonstructural protein 1) protein plays several roles in viral infections. Inside infected cells, it is essential to making new copies of the virus to infect additional cells. Infected cells also secrete NS1 packets into the patient's blood stream, where higher levels have been associated with more severe illness.
The cross-shaped protein has two distinct surfaces. The inner surface is "greasy" and is believed to interact with cell membranes, while the outer surface, once secreted into the blood, can interact with the patient's immune system.
Even without the virus present, the secreted version of some NS1 proteins can create vascular bleeding, such as is seen in severe dengue infections.
X-ray crystallography data was collected at the National Institute of General Medical Sciences and National Cancer Institute (GM/CA-XSD) facility at the U.S. Department of Energy’s Advanced Photon Source, an Office of Science user facility at Argonne National Laboratory. (Janet Smith is Scientific Director of GM/CA-XSD). The electronic microscopy was conducted by the Skiniotis lab at the LSI.
Support for the research was provided by Janet Smith’s Margaret J. Hunter Collegiate Professorship and the U-M Life Sciences Institute. The National Institute of General Medical Sciences and National Cancer Institute facility at Argonne National Laboratory has been funded in whole or in part with Federal funds from the National Cancer Institute (ACB-12002) and the National Institute of General Medical Sciences (AGM-12006).
See a brief video on this subject at: https://youtu.be/RJkPNlRH63c
See: W Clay Brown1, David L. Akey1, Jamie R. Konwerski1, Jeffrey T. Tarrasch1, Georgios Skiniotis1, Richard J. Kuhn2, and Janet L. Smith1*, “Extended surface for membrane association in Zika virus NS1 structure,” Nat. Struct. Mol. Biol., published online July 25, 2015. DOI: 10.1038/nsmb.3268
Author affiliations: 1University of Michigan, 2Purdue University
Correspondence: *[email protected]
IN ADDITION: Research at the Northeastern Collaborative Access Team 24-ID beamline at the APS produced "a high-resolution (1.62-Å) crystal structure of the RNA helicase from the French Polynesia strain [of the virus]." These results are the subject of: "Structure of the NS3 helicase from Zika virus," Rinku Jain, Javier Coloma, Adolfo García-Sastre, and Aneel K Aggarwal, Nat. Struct. Mol. Biol. Brief Communication, advance publication, published online 11 July 2016. DOI: 10.1038/nsmb.3258
Read: “This giant X-ray machine helped decode one of the Zika virus' secrets,” Chicago Tribune, July 27, 2016.
Hear: “New Zika Research: Key Virus Protein Structure Solved,” “The 21st Show” report, including interviews with Janet Smith, and Argonne Associate Laboratory Director and APS Director Stephen Streiffer.
Watch: “Ground breaking Zika research performed in suburb,” WGN-TV report, including interviews with Janet Smith and Robert Fischetti, Life Sciences Advisor to the APS Director and Group Leader, GM/CA-XSD.
Argonne National Laboratory is supported by the Office of Science of the U.S. Department of Energy. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States, and is working to address some of the most pressing challenges of our time. For more information, please visit science.energy.gov.