The original UC San Diego Health System press release by Heather Buschman can be read here.
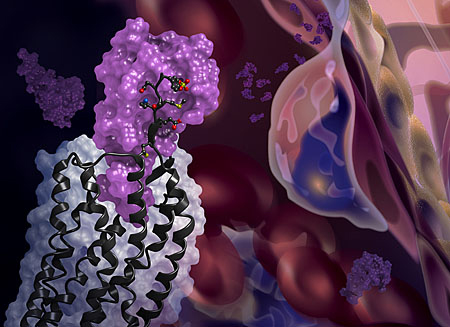
The first crystal structure of the cellular receptor CXCR4 bound to an immune signaling protein called a chemokine has been determined by researchers using high-brightness x-rays from the U.S. Department of Energy’s Advanced Photon Source (APS). The structure, published Jan. 22 in Science, answers longstanding questions about a molecular interaction that plays an important role in human development, immune responses, cancer metastasis and HIV infections.
“This new information could ultimately aid the development of better small molecular inhibitors of CXCR4-chemokine interactions — inhibitors that have the potential to block cancer metastasis or viral infections,” said Tracy M. Handel, professor of pharmacology at UC San Diego and senior author of the study.
CXCR4 is a receptor that sits on the outer surface of cells, sticking out like an antenna. When it receives a message, in the form of signaling molecules called chemokines, the receptor binds the chemokines and transmits the message to the inside of the cell. This signal relay helps cells migrate normally during development and inflammation. But CXCR4 signaling can also play a role in abnormal cell migration, such as when cancer cells metastasize. CXCR4 is infamous for another reason: HIV uses it to bind and infect human immune cells.
Despite its far-reaching consequences, researchers have long lacked data to show how exactly the CXCR4-chemokine interaction occurs, or even how many CXCR4 receptors a single chemokine molecule might simultaneously engage. This is because membrane receptors like CXCR4 are exceptionally challenging structural targets. The difficulty dramatically increases when studying such receptors in complexes with the proteins they bind.
To overcome these experimental challenges, Handel’s team of researchers from the University of California, San Diego, and The Scripps Research Institute used a novel approach. They combined computational modeling and a technique known as disulfide trapping to stabilize the complex. Once stabilized, the researchers were able to use x-ray crystallography at the National Institute of General Medical Sciences and National Cancer Institute (GM/CA-XSD) 23-ID-B and 23-ID-D x-ray beamlines at the APS to determine the CXCR4-chemokine complex’s three-dimensional atomic structure. (The APS is an Office of Science user facility.)
This is the first time that a receptor like CXCR4 has been crystallized with a protein binding partner and the results revealed several new insights. First, the new crystal structure shows that one chemokine binds to just one receptor. Additionally, the structure reveals that the contacts between the receptor and its binding partner are more extensive than previously thought — it is one very large contiguous surface of interaction rather than two separate binding sites.
“The plasticity of the CXCR4 receptor — its ability to bind many unrelated small molecules, peptides and proteins — is remarkable,” said Irina Kufareva, a computational scientist at UC San Diego and co-corresponding author of the study. “Our understanding of this plasticity may impact the design of therapeutics with better inhibition and safety profiles.”
“With more than 800 members, seven-transmembrane receptors like CXCR4 are the largest protein family in the human genome,” added Raymond Stevens, provost professor and director of the Bridge Institute at the University of Southern California and co-corresponding author. “Each new structure opens up so many doors to understanding different aspects of human biology, and this time it is about chemokine signaling.”
See: Ling Qin1, Irina Kufareva1***, Lauren G. Holden1*, Chong Wang2, Yi Zheng1, Chunxia Zhao1, Gustavo Fenalti2, Huixian Wu2, Gye Won Han2, Vadim Cherezov2‡, Ruben Abagyan1, Raymond C. Stevens2**‡, and Tracy M. Handel1, “Crystal structure of the chemokine receptor CXCR4 in complex with a viral chemokine,” Science Express, published online January 22, 2015. DOI: 10.1126/science.1261064
Author affiliations: 1University of California, San Diego 2The Scripps Research Institute
Present address: ‡Present address: Bridge Institute, University of Southern California, Los Angeles, CA 90089
Correspondence: * [email protected], ** [email protected], *** [email protected]
This research was made possible by the PSI:Biology program funded by the National Institute of General Medical Sciences at the National Institutes of Health (NIH). This research was also funded, in part, by NIH grants R01GM071872, U01GM094612, R01GM081763, R21AI101687, U54GM094618, Y1-CO-1020 and Y1-GM-1104, and the Pharmaceutical Research and Manufacturers of America Foundation. L.G.H. is supported by a 2012 Postdoctoral Fellowship in Pharmacology/Toxicology from the Pharmaceutical Research and Manufacturers of America (PhRMA) Foundation. GM/CA-XSD has been funded in whole or in part with Federal funds from the National Cancer Institute (ACB-12002) and the National Institute of General Medical Sciences (AGM-12006). This research used resources of the Advanced Photon Source, a U.S. Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under Contract No. DE-AC02-06CH11357.
Argonne National Laboratory is supported by the Office of Science of the U.S. Department of Energy. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States, and is working to address some of the most pressing challenges of our time. For more information, please visit science.energy.gov