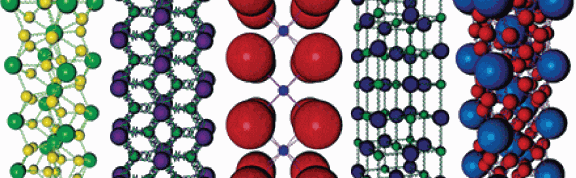
Nature is a master builder. Using a bottom-up approach, nature takes tiny atoms and, through chemical bonding, makes crystalline materials, like diamonds, silicon, and even table salt. In all of them, the properties of the crystals depend upon the type and arrangement of atoms within the crystalline lattice.
Now, Northwestern University scientists, with help from an x-ray beamline at the U.S. Department of Energy Office of Science’s Advanced Photon Source, have learned how to top nature by building crystalline materials from nanoparticles and DNA, the same material that defines the genetic code for all living organisms.
Using nanoparticles as "atoms" and DNA as "bonds," the scientists have learned how to create crystals with the particles arranged in the same types of atomic lattice configurations as some found in nature, but they also have built completely new structures that have no naturally occurring mineral counterpart.
The basic design rules the Northwestern scientists have established for this approach to nanoparticle assembly promise the possibility of creating a variety of new materials that could be useful in catalysis, electronics, optics, biomedicine and energy generation, storage and conversion technologies.
"We are building a new periodic table of sorts," said Northwestern’s Chad A. Mirkin, who led the research. "Using these new design rules and nanoparticles as 'artificial atoms,' we have developed modes of controlled crystallization that are, in many respects, more powerful than the way nature and chemists make crystalline materials from atoms. By controlling the size, shape, type and location of nanoparticles within a given lattice, we can make completely new materials and arrangements of particles, not just what nature dictates."
"Once we have a certain type of lattice," Mirkin said, "the particles can be moved closer together or farther apart by changing the length of the interconnecting DNA, thereby providing near-infinite tunability."
"This work resulted from an interdisciplinary collaboration that coupled synthetic chemistry with theoretical model building," said coauthor George C. Schatz, also at Northwestern. "It was the back and forth between synthesis and theory that was crucial to the development of the design rules. Collaboration is a special aspect of research at Northwestern, and it worked very effectively for this project."
In the study, the researchers started with two solutions of nanoparticles coated with single-stranded DNA. They then added DNA strands that bound to these DNA-functionalized particles, which then presented a large number of DNA "sticky ends" at a controlled distance from the particle surface; these sticky ends then bound to the sticky ends of adjacent particles, forming a macroscopic arrangement of nanoparticles.
Different crystal structures are achieved by using different combinations of nanoparticles (with varying sizes) and DNA linker strands (with controllable lengths). After a process of mixing and heating, the assembled particles transitioned from an initially disordered state to one where every particle was precisely located according to a crystal lattice structure. The process is analogous to how ordered atomic crystals are formed.
The researchers report six design rules that can be used to predict the relative stability of different structures for a given set of nanoparticle sizes and DNA lengths. In the paper, they use these rules to prepare 41 different crystal structures with nine distinct crystal symmetries. However, the design rules outline a strategy to independently adjust each of the relevant crystallographic parameters, including particle size (varied from 5 to 60 nanometers), crystal symmetry, and lattice parameters (which can range from 20 to 150 nanometers).
The lattices were characterized via small-angle x-ray scattering experiments performed on the DuPont-Northwestern-Dow Collaborative Access Team (DND-CAT) beamline 5-ID, Advanced Photon Source, Argonne National Laboratory. Accurately analyzed structural data is crucial in moving this type of research to the next step. The APS provides high flux and a well-collimated x-ray beam, which enables in situ measurement while annealing samples.
These 41 crystals are just a small example of the near infinite number of lattices that could be created using different nanoparticles and DNA strands.
Mirkin and his team used gold nanoparticles in their work but note that their method also can be applied to nanoparticles of other chemical compositions. Both the type of nanoparticle assembled and the symmetry of the assembled structure contribute to the properties of a lattice, making this method an ideal means to create materials with predictable and controllable physical properties.
Mirkin believes that, one day soon, software will be created that allows scientists to pick the particle and DNA pairs required to make almost any structure on demand.
See: Robert J. Macfarlane1, Byeongdu Lee2, Matthew R. Jones1, Nadine Harris1, George C. Schatz1, Chad A. Mirkin1,2*, “Nanoparticle Superlattice Engineering with DNA,” Science 334, 204 (14 OCTOBER 2011). DOI:10.1126/science.1210493
Author affiliations: 1Northwestern University, 2Argonne National Laboratory
Correspondence: *[email protected]
This work was supported by the Defense Research & Engineering Multidisciplinary University Research Initiative of the Air Force Office of Scientific Research, and by the U.S. Department of Energy Office of Basic Energy Sciences [award DE-SC0000989; Northwestern University (NU) Non-equilibrium Energy Research Center] (C.A.M. and G.C.S.); a National Security Science and Engineering Faculty Fellowship from the U.S. Department of Defense (C.A.M.); an NU Ryan Fellowship (R.J.M.); and an NU Ryan Fellowship and a National Science Foundation Graduate Research Fellowship (M.R.J.). DND-CAT is supported by E. I. DuPont de Nemours & Co., The Dow Chemical Company, and Northwestern University.
Use of the APS was supported by the U.S. Department of Energy Office of Science under contract DE-AC02-06CH11357.
The “Science Perspective” article entitled, “Self-Assembly Enters the Design Era,” by Alex Travesset (Ames Lab) can be read here.
The original Northwestern University press release by Megan Fellman can be read here.
Audio of Chad Mirkin discussing this research on the National Public Radio program “Talk of the Nation”. Video of a similar discussion by Mirkin is available upon request by contacting Lisa-Joy Zgorski at the National Science Foundation, 703-292-8311 or [email protected].
The Advanced Photon Source at Argonne National Laboratory is one of five national synchrotron radiation light sources supported by the U.S. Department of Energy’s Office of Science to carry out applied and basic research to understand, predict, and ultimately control matter and energy at the electronic, atomic, and molecular levels, provide the foundations for new energy technologies, and support DOE missions in energy, environment, and national security. To learn more about the Office of Science x-ray user facilities, visit http://science.energy.gov/user-facilities/basic-energy-sciences/.
Argonne National Laboratory seeks solutions to pressing national problems in science and technology. The nation's first national laboratory, Argonne conducts leading-edge basic and applied scientific research in virtually every scientific discipline. Argonne researchers work closely with researchers from hundreds of companies, universities, and federal, state and municipal agencies to help them solve their specific problems, advance America's scientific leadership and prepare the nation for a better future. With employees from more than 60 nations, Argonne is managed by UChicago Argonne, LLC for the U.S. Department of Energy's Office of Science.